Unlock monolithic clean in place and steam in place procedures to optimize process KPIs, OEE and profits. Batch automation model, control programming, HMIs can help.
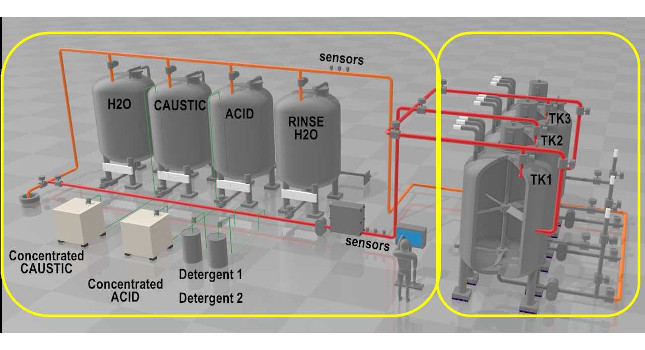
Process plant operators seeking to automate their manufacturing processes should consider automating clean in place (CIP) and sterilize in place (SIP) sequences, which often are more complex than making the products (see Figure 1). Attention to batch automation models, programming, and human-machine interface design can help, in one example improving production capacity by 20%. Some of the complexities may come from the lack of automated devices, as well as creating control code to perform the procedures defined during automation design, then, changing them until they meet the final qualifications. CIP and SIP automation are commonly prescribed with little flexibility that often only allows the end user to change minor functionality via parameter sets. Sometimes the sequences are “black box” where nothing can be viewed or changed, which limits the end user from making significant optimization changes without requiring control system reprograming.
When specifying the optimal CIP and SIP sequence, users may need to consider the many factors that affect equipment cleaning and sterilization, such as the surface characteristics, environmental considerations, chemical factors, detergent concentration, temperature, chemical active ingredients, physical active ingredients, chemistry of detergents, acid-anionic sanitizers, hydrogen peroxide, exposure time, flow rate and so on.
Changing the paradigm of how CIP and SIP are implemented is the key to unlocking the potential for improving many aspects that can help optimize them and therefore optimize the overall equipment effectiveness (OEE) of the process. A major aspect of automation is to provide the ability to clearly specify how the procedures should be executed, enforce their execution and provide ample information for verifying how the activities were performed, while the system should allow the process to be optimized regardless of whether these activities are executed by the automated equipment or via operator actions.
Operators can clearly be instructed to perform the required tasks at the appropriate time. These tasks may consist of equipment setup configuration images or manual material addition instructions while capturing pertinent information.
This optimization capability can be achieved with off-the-shelf products and a paradigm change in the implementation approach. The system must provide the maximum flexibility and modularity based on the equipment capabilities, as well as incorporate the required operator actions as part of these procedures.
Procedure automation does not imply that the equipment is automated. It means that all steps and actions required to perform all tasks are executed in the correct order in an unambiguous way regardless of how these tasks are executed (automated equipment or operator actions).
Often, the automation solution assumes the operators have performed the required setup activities prior to starting the actual CIP and SIP automated sequence. Unfortunately, the workforce does not always perform these activities consistently. Even experienced operators tend to have their own way of doing things, and the new operator may be at risk of not performing the required activities as intended. Therefore, a comprehensive solution should incorporate all the activities the control system can execute as well as the activities the operators must perform, such as making connections on transfer panels, removing equipment and so on.
Improving CIP and SIP procedures
To improve CIP and SIP procedures, expose capabilities of the equipment and personnel to a recipe author who will be responsible for configuring the required procedures without requiring the intervention of the automation team. The best practices as to how to perform CIP and SIP can be specified and modified without requiring controller programming. Due to this implementation approach, an end user can continuously update the current best approach and improve OEE.
In addition to walking the operators and automated equipment through the required cleaning and sterilizing steps, these off-the-shelf products provide the ability to arbitrate equipment and recipe execution of the products as well as CIP and SIP. They maintain and update equipment status (clean, in use, to be cleaned, out of service, in service and the like); the recipe author can confidently manage the recipes through their lifecycle via versioning as well as recipe approval processes. Since the solution manages the execution of the products, as soon as the equipment can be cleaned or sterilized, the procedures can commence without requiring the operator to initiate them. Equally, the product recipes can start as soon as the required equipment CIP or SIP procedures have completed.
Modular approach programming
A modular approach to automating the process can best be obtained by following the ISA-S88 standards for batch automation, where a fundamental concept is to expose equipment capabilities to a recipe author. The ISA-S88 equipment model exposes these capabilities. The ISA-S88 procedural model is used by the recipe author to specify how these capabilities are used. The equipment capabilities often are programed in controllers and the procedures are configured as required by the recipe author via a PC display, not a programming tool.
The procedural model is shown on the left side of Figure 2; levels ISA-S88 defines in the equipment model are shown on the right side of Figure 2. Starting at the bottom within the phase, the basic procedural model building block, for every equipment phase defined, there is a representation of it in the procedural model.
This implementation philosophy provides the maximum exposure of the equipment capabilities. It is important not to focus on how the recipes can be implemented but more on what the equipment can be commanded to do. It is important to expose the capabilities of what can be done by the CIP skid and SIP equipment and at the destination equipment.
Figure 3 shows a control system human-machine interface (HMI) equipment module faceplate that represents what the CIP skid and the destination equipment can be commanded to do. The operator can control the equipment module via the HMI, or the sequencing engine can control it in automatic mode. The operator can specify the parameters to complete a minor step of the CIP procedure. Once this is complete, the operator can configure the next step and so on until all the steps are complete. But a recipe procedure can be configured to perform the entire sequence of steps automatically until the equipment is cleaned or sterilized using the same concept but commanded by the sequencing engine.
Commands on the left side of the equipment module faceplate allows users to:
-
Deliver a material from a source
-
Control the flow rate
-
Control the temperature
-
Receive material and return it to a desired route
-
Monitor supply and return conditions.
The logic also monitors required conditions to determine if the desired duration and/or amount required has been met.
The destination equipment capabilities are visible on the right side: Receive and return CIP material via possible flow paths.
These parameters can be populated from a recipe for each step and the process conditions monitored by the recipe to make decisions regarding when to proceed to the next recipe step. The recipe author is exposed to process information for configuring recipe transition conditions such as supply and return conductivity, flow rate, temperature as well as the destination information such as temperature, level, pressure and any other equipment information available.
The operator can manually control independent CIP and SIP configurations via the HMI faceplate, but the idea is to have the sequencing engine drive all the activities as specified in the recipe procedures. Following the ISA-S88 standard, we implement equipment phases — much like building blocks — that can take the role of the operator and specify how this equipment module should be used. This equipment phase is then called by the recipe author to transfer the desired parameters to the equipment module as many times and in as many ways as necessary to perform the current best approach for cleaning and sterilizing a piece of equipment.
Recipe phases, the building blocks on the procedural side, are put together to form “operations.” Multiple operations can be created to maximize reuse of operations across multiple recipes. An operation can be created to prompt the operator to prepare the equipment for CIP or SIP, another operation can be created to perform a quick rinse or a comprehensive clean based on the condition of the equipment that requires cleaning or sterilizing. These operations are organized and sequenced inside of a unit procedure as seen in Figure 4, unit procedures can be sequences within a procedure level recipe.
The key to simplifying the CIP and the SIP process is to set up equipment using an equipment module, this module being able to define all possible equipment configurations in a simple modular matter. Modular, straightforward controller design and implementation approach, coding requirements consist of simple to define and implement actions, such as open paths, control flow rate, control temperature and expose process conditions. What to do with these capabilities is the role of the recipe/procedure author. Application of the procedural role of ISA-S88 enables the specification of the sequence necessary to perform the required procedures.
Some of the benefits of this approach include:
-
Recipe author focuses on specifying the procedures to be followed, not the code.
-
Automation team focuses on exposing the equipment capabilities to the recipe editor.
-
No need to change the controller code to improve procedures.
-
Create reusable operations to deploy across multiple standard operating procedures.
-
Incorporate manual procedures within the overall procedures.
-
Provide new and experienced operator a consistent methodology for preparing equipment for CIP and SIP.
-
Provide unambiguous instructions and information to properly setup each manual step.
-
Create consistency by enforcing execution steps.
-
Capture procedural information for reporting and analytics.
-
Capture OEE information for each step, as well as group of tasks.
-
Flexible and modular automation-enabled continuous improvements.
CIP and SIP constitute a critical aspect of the bottom line of a manufacturing process. Just as important as the products manufactured with the assets, CIP plays a very important role in the product quality as well as the overall availability of the equipment to manufacture these products. Being able to consistently execute the required cleaning step regardless of the equipment level of automation is paramount, therefore being able to clearly specify and enforce the steps required are critical regardless of how these tasks are executed. Operators and equipment are guided to seamlessly perform their task to ensure the desired cleaning procedures are executed in an optimum manner.
Batch optimization: 20% capacity gain
The heart of agile cleaning is an ISA-S88 batch management package where cleaning requirements can be defined in one or more recipes rather than in the control programming and data tables. Here we find the first advantage, that a process knowledgeable person can modify the cleaning practices without being a controller programmer or hiring one. Modifying ISA-S88 flowchart-style recipes is easy to learn and do well in comparison to controller programming.
Efficient cleaning is doing the right cleaning based on product made and equipment status. Hard work enabled by agile tools can make a dramatic difference. One client increased process cell capacity by 20% through careful experimenting, swabbing, testing and adjusting of its recipes. Because we exposed the capabilities of the equipment, they were able to modify the recipe to attempt cleaning equipment in parallel, adjust times, monitor various conditions and the like. Capacity to make 20% more product with no incremental equipment cost and minimal marginal labor cost increases plant profitability dramatically.
Not all products require the same level of cleaning. Some may require significantly less. Scheduling a cleaning recipe is designed specifically for the cleaning requirements of that product. Compared to using a single, worst-case scenario, cleaning recipe following all product runs, intelligently doing the right cleaning increases available process cell capacity and plant profits.
Using a batch management package for cleaning recipes provides an electronic record of the cleaning. Such records can be used in a variety of ways to improve cleaning practices and to identify cleaning. Building from the previous example, a monitoring solution can validate from the electronic record that the right cleaning recipe was executed and completed without anomalies. Admittedly, lowered risk only affects plant profitability indirectly, but is certainly valuable to business overall.
This agile approach was used on a 60-unit process cell initially designed so that any unit might be cleaned by any of three cleaning systems. After being in production for several months, the client recognized the value of investing in a fourth cleaning system, allowing parallel cleaning of four units instead of three. This change could be supported in approximately a day of engineering time with essentially no startup time as new supply and return equipment modules were added with a fourth coordinator capable of using the existing 60 process supply and 60 process return equipment modules. The additional cleaning system improved the process cell OEE, increased cell capacity and improved plant profitability.
These examples apply to both CIP and SIP practices. Recipe procedures in a batch management package can easily direct operators to perform manual actions.
Implementing agile ISA-S88 solutions for batch process control and cleaning can cost 10% to 20% more initially. However, the investment can easily pay large dividends over the lifecycle of the project.
John Parraga is a batch process specialist at ECS Solutions. He is an experienced batch process engineer with career stops at Sequentia and Rockwell Automation. ECS Solutions is a certified member of the Control System Integrators Association (CSIA). Edited by Chris Vavra, associate editor, Control Engineering, CFE Media and Technology, [email protected].
Learn more about ECS Solutions in the Global System Integrator Database.



